Reversed-phase chromatography (RP-HPLC) is the foundation of modern chemical analysis, accounting for over 80% of all separations performed in R&D, quality control and clinical diagnostics laboratories. Its dominance stems from its versatility, the stability of stationary phases and its excellent compatibility with mass detection (LC-MS).

Physicochemical retention mechanism
In the article “Fundamentals of chromatography“, we have already explained the difference between normal phase and reversed phase chromatography. However, it is worth returning to this topic. It concerns the arrangement of phases (mobile and stationary). In normal phase chromatography, the stationary phase is polar and the mobile phase is non-polar (strong organic solvents). As the name suggests, in reverse phase chromatography, it is exactly the opposite. The stationary phase is non-polar (hydrophobic) and the mobile phase is polar.
Interestingly, inverted phases are the most common today. This is because they are much more versatile.
Mechanism: Why do substances “escape” into the column?
Most people think that the column (stationary phase) acts like a magnet that captures molecules. In reality, the opposite is true:
- Water likes water: Water molecules in the liquid flowing through the column (mobile phase) are very strongly bonded to each other (forming a network of hydrogen bonds).
- The analyte is an intruder: When we inject a substance (analyte) that does not like water, it ‘pushes’ the water molecules apart. This is very tiring and energetically disadvantageous for the system.
- Pushing out: Water wants to regain its calm as quickly as possible, so it literally pushes the intruder towards the “fat” chains lining the column. The substance hides there, just to have as little contact with water as possible.
Three main forces within the resin
Once molecule has been pushed out of the water and reaches the hydrophobic groups of the stationary phase, three types of interactions begin to take place:
1. London forces (the most important)
It is a kind of ‘molecular glue’. It occurs between the hydrophobic parts of the molecule and the hydrophobic chains in the column. The more ‘fatty’ (non-polar) the molecule is, the more strongly it sticks to the column and the slower it flows out of it.
2. Dipole-Dipole Interactions
They act a bit like small magnets. If the substance and the column have specific chemical groups (e.g. phenyl or cyanide), they can additionally “stick” to each other.
3. Electrostatic Interference (Often problematic)
It’s pure electricity. Acid groups (e.g. silane groups) are sometimes exposed on the surface of the column. If the analyte has a positive charge, it will “stick” to them too strongly. Chemists usually try to avoid this because it causes peak tailing.
In short: Water does not like non-polar analytes, so it pushes them to the side (to the column). How long the substance stays there depends on how much it likes the hydrophobic column filling and how we control the polarity of the mobile phase.
Characteristics of stationary phases and column construction
Wybór odpowiedniej kolumny jest krytycznym etapem projektowania metody. Wydajność separacji zależy zarówno od chemii powierzchni, jak i fizycznej struktury nośnika.
Nośnik krzemionkowy i jego modyfikacje
Most RP-HPLC columns are based on ultra-pure type B silica (with low metal content). Key parameters are:
- Particle size (dp): Typically 3–5 μm for HPLC, less than 2 μm for UHPLC. Reducing the particle diameter dramatically increases column efficiency (N), but generates high back pressure (see also: introduction to HPLC).
- Pore size: Standard 60–120 Å for small molecules. For proteins and large peptides, wide-pore silica (300 Å) is used to allow free diffusion of macromolecules into the pores.
Types of functional groups
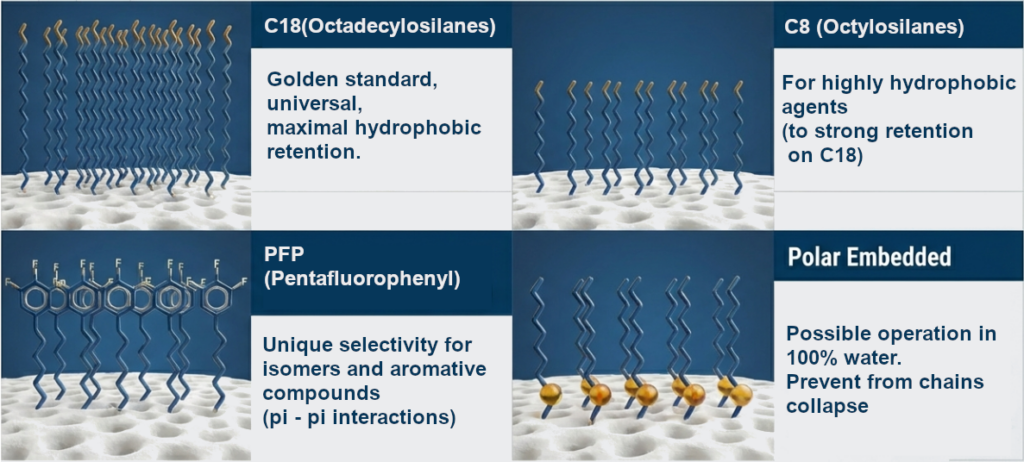
- C18 (Octadecylosilanes): The most versatile phase, providing maximum hydrophobic retention.
- C8 (Octylsilanes): Used for highly hydrophobic compounds where C18 retention would be too strong.
- PFP (Pentafluorophenyl): Offer unique selectivity for isomers and aromatic compounds due to pi-pi and dipolar interactions.
- Polar Embedded phases: These contain a polar group (e.g. amide) embedded in an alkyl chain. They allow operation in a 100% aqueous phase without phase collapse.
End-capping: Why is it essential?
During silica modification, for steric reasons, not all silanol groups (-OH) bind to the C18 ligand. Free silanols are acidic and can cause peak tailing, especially for amines. End-capping is the process of blocking these groups with small molecules, e.g. trimethylchlorosilane (TMS), which significantly improves peak symmetry and column chemical stability.
Distribution optimisation parameters
Success in RP-HPLC depends on precise tuning of the mobile phase parameters.
Composition of the mobile phase
The polarity of the mobile phase can be controlled. Organic modifiers are most commonly used for this purpose: acetonitrile (ACN) and methanol (MeOH). Adding them to the mobile phase or gradually increasing their concentration (see the gradient section) affects the retention time of hydrophobic analytes.
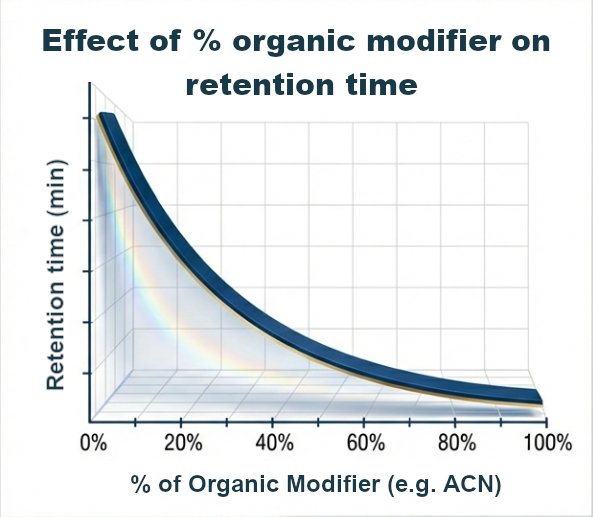
ACN has lower viscosity and higher elution power than MeOH. MeOH may offer different selectivity due to its ability to form hydrogen bonds. The choice of solvent is one of the things to consider when selecting an analytical method. For complex matrices, this can affect the number of peaks separated and thus the ability to detect different substances.
Retention coefficient
The retention coefficient in liquid chromatography is the ratio of the time the analyte spends in the stationary phase to the time spent in the mobile phase. It is determined from the retention time and dead volume time.
The retention coefficient k’ is described by the formula:
Where tr is the analyte retention time and t0 is the dead time. k’ should be in the range of 1–10. For quantitative analysis, it should be between 2 and 3.
A k’ value that is too low (<1) means that the analyte has poor binding to the column. Typically, peaks tail or are poorly separated. In this case, you can try to reduce the organic phase fraction at the beginning of the separation, flatten the gradient, or switch to isocratic separation. In extreme cases, you can try using a different stationary phase or a different analytical technique (e.g. HILIC).
Too high k’ (>10) prolongs the analysis time and peaks often tail. In this case, a strong gradient or even isocratic separation at a high concentration of the organic phase may help. If this happens on C18 column, try C8, which is better for highly hydrophobic agents. In extreme cases, changing the separation technique, e.g. to HIC, may help.
The role of pH and buffers
pH control is crucial for ionisable compounds.
- Acids: At low pH (pH < pKa), they are in an undissociated (neutral) form, which drastically increases their retention in RP-HPLC.
- Bases: At high pH (pH > pKa), they become neutral and retard better.
However, it is important to keep in mind the limitations of silica: at pH < 2, hydrolysis of siloxane bonds occurs, and at pH > 8, the silica skeleton dissolves.
Please note! Modern hybrid columns have a higher pH tolerance. They can sometimes operate in alkaline conditions reaching up to pH 12. Before using the column, check its operating range.
Ion-pairing agents
For highly polar, ionised molecules (e.g. small peptides) that are poorly retained, additives such as trifluoroacetic acid (TFA) or formic acid are used. TFA forms a neutral ion pair with the analyte, which increases its hydrophobicity and improves peak shape. Formic acid is preferred in LC-MS due to lower ion suppression.
Isocratic vs. gradient elution
- Isocratic: Fixed mobile phase composition. Simpler, does not require column conditioning time, but ineffective for samples with a wide range of hydrophobicity.
- Gradient: The percentage of the organic modifier increases over time. Essential in proteomics, for the analysis of pharmaceutical contaminants and complex samples.
Isocratic analysis works well for fairly simple samples. Gradient elution is ideal for complex matrices and when a single sample contains compounds with different degrees of polarity.
Please note! If you are conducting an isocratic analysis, it is worth considering the so-called cleaning gradient. It involves increasing the proportion of the organic (non-polar) phase at the end of the method to flush out any hydrophobic contaminants that may remain on the column and carry over to subsequent analyses.
Advantages and limitations of reversed-phase chromatography
Reverse-phase chromatography dominates among modern liquid chromatography techniques. It is very versatile, but like any technique, it has its limitations. Below we present the advantages and limitations of RP-LC.
| Advantages | Limitations |
| Compatibility with MS: Most mobile phases (ACN, water, volatile buffers such as ammonium acetate) can be directly introduced into the mass spectrometer. Predictability: Retention correlates with the logP of the analyte (octanol/water partition coefficient). Stability: When the appropriate care is taken, RP columns are mechanically and chemically stable. | Strongly polar compounds: Sugars or small organic acids may elute during dead time (t0). They require HILIC or ion exchange techniques. pH stability: Standard columns have a narrow operating range (pH 2–8). The solution is to use more expensive hybrid (organic-inorganic) columns. |
Practical applications of reversed-phase chromatography
Pharmacy and Quality Control
RP-HPLC is the USP/Ph. Eur. standard for testing the purity of active pharmaceutical ingredients (APIs). It allows the separation of the main product from degradation products with almost identical structures.
Biotechnology: Protein and Peptide Analysis
In therapeutic protein purification processes, RP-HPLC is used for peptide mapping. After digesting the protein with trypsin, the resulting “fingerprint” in the ACN gradient allows confirmation of the correct amino acid sequence and identification of post-translational modifications (PTM).
Forensic Science and Toxicology
Testing for the presence of xenobiotics in body fluids. Thanks to high-resolution RP-HPLC combined with MS/MS, it is possible to simultaneously detect hundreds of drugs and their metabolites in a single analysis.
Summary and conclusions
Reverse-phase chromatography is a mature but still evolving technique. Understanding that retention is a dynamic process, dependent on a delicate balance between the hydrophobicity of the analyte and the composition of the mobile phase and surface chemistry, allows for informed method design.
In the era of UHPLC and rapid separations, temperature optimisation (reducing phase viscosity) and reagent purity are becoming particularly important, as they directly translate into sensitivity and repeatability of results on an R&D scale.
Literature
- https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Supplemental_Modules_(Analytical_Chemistry)/Instrumentation_and_Analysis/Chromatography/Chromatographic_Columns
- Horváth, C., Melander, W., & Molnár, I. (1976). Solvophobic interactions in liquid chromatography with nonpolar stationary phases. Journal of Chromatography A, 125(1), 129-156.
- United States Pharmacopeia (USP). General Chapter <621> Chromatography.
- Waters Corporation (2023). Beginner’s Guide to HPLC & RP-HPLC Technology. Technical Note.
- Agilent Technologies (2022). High Performance Liquid Chromatography: Method Development Guide. Application Compendium.
